 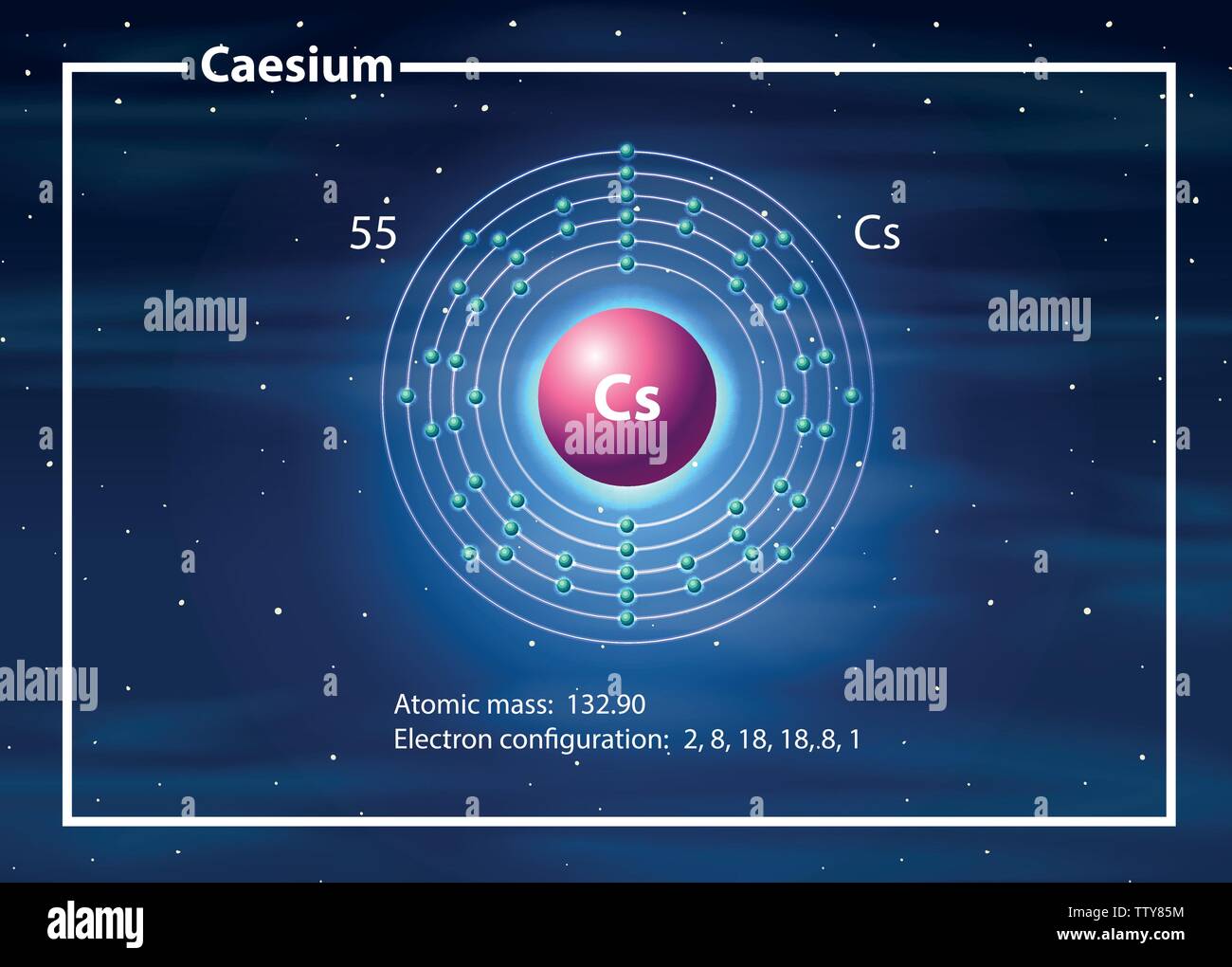
The chloride is used in photoelectric cells, in optical instruments, and in increasing the sensitivity of electron tubes. Cesium salts are used to strenght various types of glass. Cesium is sometimes used to remove traces of oxygen from the vacuum tubes and from light bulbs. Cesium nitrate is used to make optical glasses. Cesium metal oxidized rapidly when exposed to the air and can form the dangerous superoxide on its surface.Ĭesium is used in industry as a catalyst promoter, boosting the performance of other metal oxides in the capacity and for the hydrogenation of organic compounds. Cesium reacts with the halogens to form a fluoride, chloride, bromide, and iodide. Cesium hydroxide is a strong base and attacks glass. Cesium reacts explosively with cold water, and reacts with ice at temperatures above -116☌. Cesium, gallium, and mercury are the only three metals that are liquid at or around room temperature. It is the most electropositive and most alkaline element. The metal is characterised by a spectrum containing two bright lines in the blue (accounting for its name). 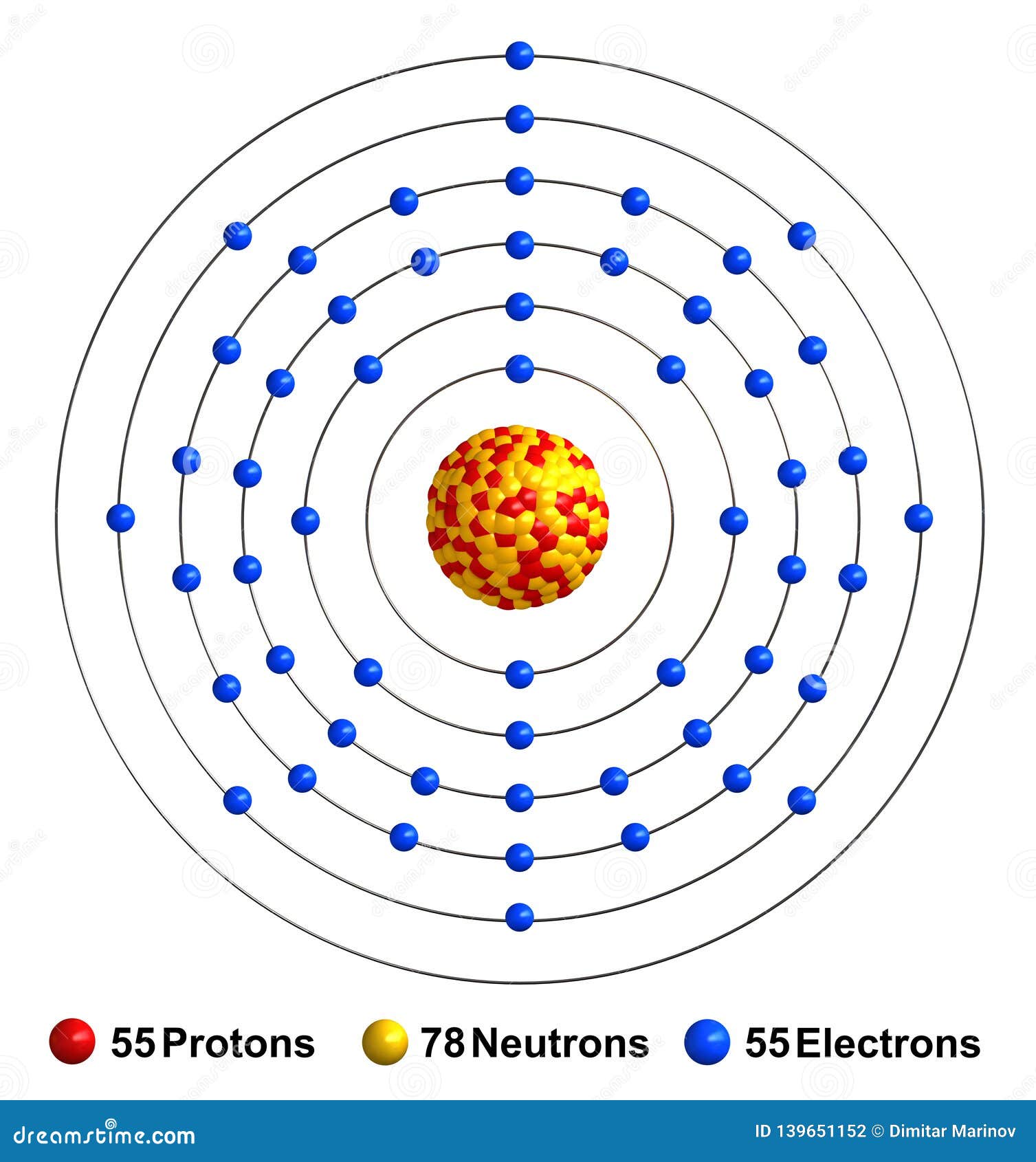
Separation and Concentration Purification RequestĬesium - Cs Chemical properties of cesium - Health effects of cesium - Environmental effects of cesium.Plant Inspection & Process Optimalisation.But this is not true if we try to compare atoms from different parts of the periodic table. This is true if we talk about atoms in the same group. Note:Some students might think atoms will get bigger if we add more shells and might conveniently ignore the other effect (the attraction between outer electrons and nucleus). 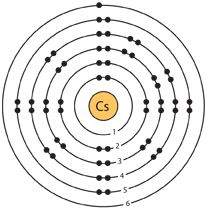
Since, in group $1$ Lithium \is at the top, so it has least shell and as a result least size as compared to caesium \ which is placed last in group and so it has largest number of shell and hence biggest size. The increase in shell, increases the size of the element. Now whenever we move from top to bottom in a group the number of shells of consecutive elements starts increasing. Thus, the only factor which affects the size of the atom is the number of shells containing electrons around the nucleus. Now we know that in any group element, the outer electrons feel a net pull of 1+ from the nucleus however this positive charge on the nucleus is cut down due to the screening effect of inner electrons. The attraction between the outer electrons and the nucleus The number of shells containing electrons around the nucleus We should always remember that radius of any element in the periodic table is determined by two factors, listed below 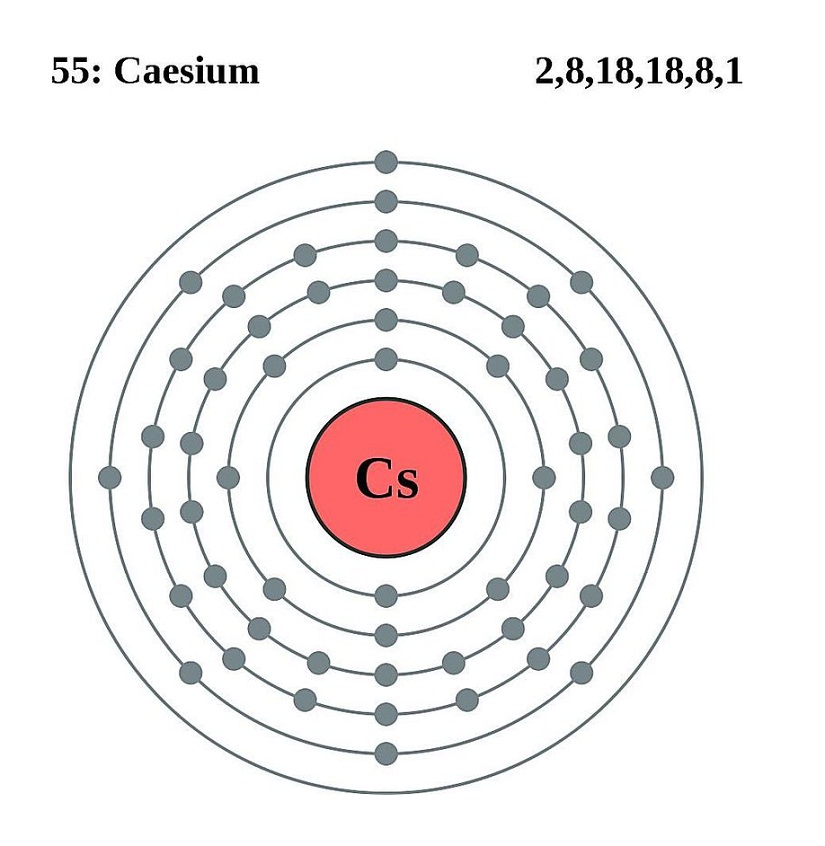
Periodicity refers to trends or recurring variation in element properties (such as atomic radii, electronegativity etc) with increasing atomic number periodicity is caused by regular and predictable variations in element atomic structure. Firstly, let us understand what periodicity means? Let us see the reason behind the statement. The same is discussed below,Ĭomplete step-by-step answer:in the question it is mentioned that Caesium \ have a larger atomic radius than Lithium \. Both \ and \ belong to group 1 and the atomic radii comparisons can be done by the regular trends we observe when we come from top to bottom in a group. Usually, it is the distance of the nucleus to the outermost shell containing electrons. Hint: Atomic radius of a chemical element in the measure of the size of its atom. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
